SancaMedChem Celebrates 20 Years of NEQUIMED
Medicinal and Biological Chemistry Group
São Carlos Chemistry Institute of the University of São Paulo
SancaMedChem Celebrates 20 Years of NEQUIMED/IQSC/USP
Monday, June 2 · 09:30 – 17:30
Time zone: America/Sao_Paulo
How to join Google Meet
Video call link: https://meet.google.com/rcg-vahz-eek
The Medicinal and Biological Chemistry Group, Institute of Chemistry of São Carlos, University of São Paulo
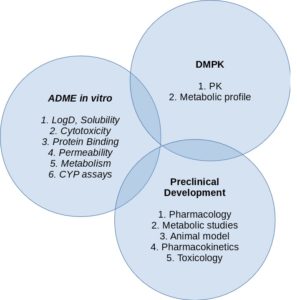
In 2025, the Medicinal and Biological Chemistry Group (NEQUIMED) proudly celebrates two decades of scientific excellence, innovation, and collaboration at the Institute of Chemistry of São Carlos (IQSC-USP). Established in 2005, NEQUIMED has become a leading research hub in Latin America, bridging chemistry and biology to discover and optimize bioactive molecules with therapeutic potential. Over the past 20 years, the group has made significant contributions to drug discovery against neglected tropical diseases, cancer, and infectious diseases, integrating cutting-edge approaches in medicinal chemistry, molecular modeling, synthetic chemistry, and chemical biology. NEQUIMED has also played a crucial role in training a new generation of scientists, fostering international partnerships, and advancing translational research. This milestone is a testament to the group’s enduring commitment to scientific rigor, societal impact, and academic excellence. As we look ahead, NEQUIMED remains dedicated to pushing the boundaries of molecular innovation to address urgent health challenges and to inspire the future of medicinal chemistry in Brazil and beyond.
Agenda for the First Edition
| 02 de Junho | |
| 09:30 – 10:00 | Opening Ceremony
Twenty years of NEQUIMED Prof. Dr. Carlos Montanari Founder and Coordinator of NEQUIMED/IQSC/USP |
| 1. 10:00 – 10:40
2. 10:40 – 11:20 3. 11:20 – 12:00 |
1. Crystallography of macromolecules: structural biology from the perspective of X-rays
Dr. William B. Fernandes 2. Hydrogen bond donor-acceptor asymmetries in drug design Dr. Peter Kenny 3. Evaluation of the protonation state of the catalytic Cys25 of cruzain as a target for Chagas disease Prof. Dr. Jerônimo Lameira |
| 12:00 – 13:30 | Almoço |
| 4. 14:00 – 14:40
5. 14:40 – 15:20 6. 15:20 – 16:00 |
4. Investigating the Lack of Translation from Cruzain Inhibition to Trypanosoma cruzi Activity with Machine Learning and Chemical Space Analyses
M.Sci. Rafael Lameiro 5. Challenges of heterologous protein production in E. coli Profa. Dra. Fernanda Canduri 6. Novel Chemical Entities Targeting Cysteine Proteases Luiz Fernando B. Da Silva |
| 16:00 – 17:30 | In-person get-together by membership |